Gruber A, Lell CP, Spruth M, Lass-Florl C, Speth C, Stoiber H, Hube B, Coleman D, Polonelli L, Dierich MP, Wurzner R (2003): HIV-1 and its transmembrane protein gp41 bind to different Candida species modulating adhesion
FEMS Immunol. Med. Microbiol. 37: 77-83.
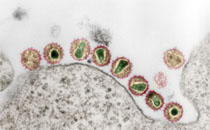
Oral candidiasis in HIV-1-infected individuals is widely believed to be triggered by the acquired T-lymphocyte immunodeficiency. Recently, binding of the HIV-1 envelope protein gp160 and its subunit gp41, and also of the whole virus itself, to Candida albicans has been shown. The present study shows that, in addition to C. albicans, HIV-1 gp41 also binds to yeast and hyphal forms of Candida dubliniensis, a species which is closely related to C. albicans, and to Candida tropicalis but not to Candida krusei, Candida glabrata or Saccharomyces cerevisiae. The previous finding that gp41 binding to C. albicans augments fungal virulence in vitro is supported by the observation that the yeast showed an enhanced adhesion to HIV-infected H9 cells in comparison to uninfected cells. In line with these results soluble gp41 itself reduced binding of C. albicans to both endothelial and epithelial cell lines, confirming a dominant role of the gp41 binding moiety on the surface of Candida for adhesion. Surface-associated secreted aspartic proteinases (Saps) play an important role in candidial adhesion, but are not likely to be involved in the interaction as gp41 binding to the C. albicans parental wild-type strain was comparable to that of three different isogenic Sap deletion mutants. Furthermore, gp41 binding to the yeast killer toxin-susceptible C. albicans strain 10S was not inhibitable by an anti-YKT receptor antibody. In conclusion, HIV-1 interacts with different clinically important Candida spp., and may thereby affect the outcome of the respective fungal infection.