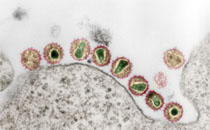
Vinjé J, Vennema H, Maunuala L, von Bonsdorff CH, Höhne M, Schreier E et al. (2003): International collaborative study to compare reverse transcriptase PCR assays for detection and genotyping of noroviruses
J. Clin. Microbiol. 41 (4): 1423-1433.
To allow more rapid and internationally standardized assessment of the spread of noroviruses (previously called Norwalk-like viruses [NLVs]) as important food-borne pathogens, harmonization of methods for their detection is needed. Diagnosis of NLVs in clinical diagnostic laboratories is usually performed by reverse transciptase PCR (RT-PCR) assays. In the present study, the performance of five different RT-PCR assays for the detection of NLVs was evaluated in an international collaborative study by five laboratories in five countries with a coded panel of 91 fecal specimens. The assays were tested for their sensitivity, detection limit, and ease of standardization. In total, NLVs could be detected by at least one RT-PCR assay in 69 (84%) of the samples that originally tested positive. Sensitivity ranged from 52 to 73% overall and from 54 to 100% and 58 to 85% for genogroup I and II viruses, respectively. In all, 64% of the false-negative results were obtained with a set of diluted stools (n = 20) that may have lost quality upon storage. Sensitivity was improved when these samples were excluded from analysis. No one single assay stood out as the best, although the p1 assay demonstrated the most satisfactory overall performance. To promote comparability of data, this assay will be recommended for newly starting groups in future collaborative studies.