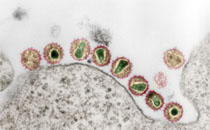
Niedrig M, Kürsteiner O, Herzog C, Sonnenberg K (2008): Evaluation of an Indirect Immunofluorescence Assay for Detection of IgM and IgG Antibodies against Yellow Fever Virus
Clin. Vaccine Immunol. 15 (2): 177-181. Epub 2007 Nov 28.
The first commercial indirect immunofluorescence assay (IFA) using Euroimmun Biochip technology was evaluated for the serodiagnosis of immunoglobulin G (IgG) and IgM antibodies against yellow fever virus (YFV) and was compared with the plaque reduction neutralization test (PRNT), which is currently the gold standard test for YFV. An overall correlation between the tests of 98.7% was established based on the analysis of 150 sera from individuals after vaccination with the 17D yellow fever vaccine. The sensitivity and specificity, calculated using the 150 sera from vaccinees and 150 sera from healthy blood donors, were 95% and 95%, respectively, for the IgG IFA and 94% and 97% for the IgM IFA. Antibody titers found in the PRNT correlated poorly with the IgM and IgG titers detected by IFA. The analysis of preexisting heterologous flaviviral immunity revealed the presence of antibodies reactive with YFV, tick-borne encephalitis virus, West Nile virus, Japanese encephalitis virus, and dengue virus serotypes 1 to 4 in 20 out of the 150 vaccinees. The indirect IFA showed that nine of these individuals with previous flaviviral exposure who received 17D vaccine failed to produce detectable IgM antibodies. Despite this preexisting immunity, all vaccinees developed protective immunity as detected by PRNT and anti-YFV IgG antibodies as detected by IFA. The high specificity and sensitivity of the IFA make it a useful tool for rapid diagnosis of yellow fever during outbreaks, for epidemiological studies, and for serosurveillance after vaccination.